Check Your Walmart Gift Card Balance: Easy Steps & Tips
Discover how to effortlessly check your Walmart gift card balance and troubleshoot common issues. Learn about the versatility of using it in-store, online, or...
Read moreEcommerce & WorkplaceAnd More
Latest News
Check Your Walmart Gift Card Balance: Easy Steps & Tips
Discover how to effortlessly check your Walmart gift card balance and troubleshoot common issues. Learn about the versatility of using...
KeySearch Review: Uncover SEO Gold & Boost Your Rankings
Unlock the secrets of SEO success with our comprehensive KeySearch review. Learn how easy it is to start, conduct keyword...
GPTDash Review: Transform Your Content Game & Unlock Creativity
Discover GPTDash, the AI content creation tool revolutionizing marketing strategies. With an easy-to-learn interface and a risk-free 30-day trial, it's...
Explaindio Review 2024: Perfect for Beginners & Pros Alike?
Discover the ultimate Explaindio review, addressing FAQs for beginners and experts alike. Learn about its user-friendly design, affordability, and versatile...
Zapable Review 2024: Unlocking the Power of Mobile Apps
Discover the power of Zapable, your go-to instant mobile app agency. Learn about pricing, exclusive bonuses, and get answers to...
Creativio AI Review: Transform Your Visuals with Ease & Creativity
Discover how Creativio AI revolutionizes visual content creation with its user-friendly interface and customization options. Learn about its universal applicability...
Activate Bankmobilevibe.com Card In 2024?
Are you encountering issues during the bankmobilevibe.com Card activation process? We are here with a step-by-step explanation of the bankmobilevibe.com...
Activate Brinksprepaidmastercard.com Card In 2024?
Encountering issues during the brinksprepaidmastercard.com Card activation process? We are here with a step-by-step explanation of the brinksprepaidmastercard.com Card activation...
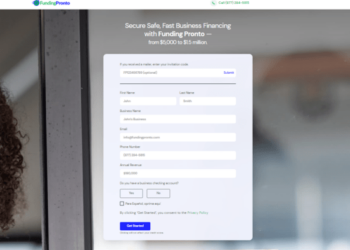
Activate Fundingpronto.com Card In 2024
Encountering issues during Fundingpronto.com Card activation process? We are here with a step-by-step explanation of the fundingpronto.com Card activation process!...
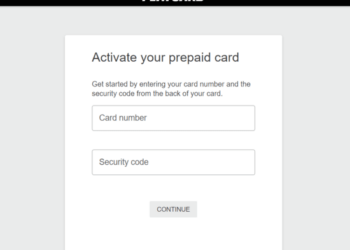
Activate Playcard.com Card In 2024?
Encountering issues during the playcard.com Card activation process? We are here with a step-by-step explanation of the playcard.com Card activation...
News Index
Check Your Walmart Gift Card Balance: Easy Steps & Tips
Discover how to effortlessly check your Walmart gift card balance and troubleshoot common issues. Learn about the versatility of using...